Furfural is an organic compound that is obtained from various agricultural byproducts  such as corncobs, oat, wheat bran, and sawdust. Furfural was first isolated by the German scientist in 1840 as a byproduct of formic acid synthesis.
such as corncobs, oat, wheat bran, and sawdust. Furfural was first isolated by the German scientist in 1840 as a byproduct of formic acid synthesis.
Furfural is a class of hetrocyclic aromatic compounds characterized by five membered ring structure with a chemical formula of C5H4O2. Furfural is an organic compound that is obtained from various agricultural byproducts such as corncobs, oat, wheat bran, and sawdust. Furfural was first isolated by the German scientist in 1840 as a byproduct of formic acid synthesis. Furfural could be produced by from a wide variety of crop materials, including corn, oats, bran, and sawdust.
Properties of furfural
- Furfural is a clear, volatile and mild toxic liquid.
- Furfural is a colorless oily liquid and its readily soluble in most of the organic solvents.
- Furfural is not a stable compound like benzene.
- At higher temperature, Furfural decomposes into Furan and carbon monoxide, that leads to an explosive reaction.
Production of Furfural Furfural is manufactured from hydrolysis of polysaccharide hemicelluloses to yield furfural compound. Hemicellulose material is available in most of the plants and easily available raw material feedstock for the production of Furfural. Furfural is produced by removing water from or dehydrating five-carbon sugars such as xylose and arabinose. These pentose sugars are commonly obtained from the hemicellulose fraction of biomass wastes like corn stalks, corncobs and the husks of peanuts & oats.
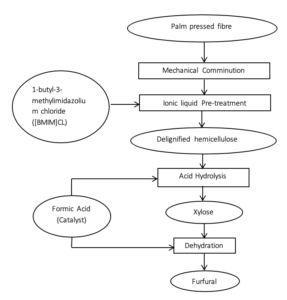
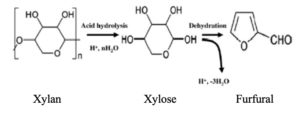
One common biomass waste is palm pressed fibre (PPF). In order to obtain furfural from PPF, lignin in hemicellulose was first removed through ionic liquid pre-treatment. Xylan then undergoes acid hydrolysis with formic acid as a catalyst to form Xylose. Furfural is finally formed from dehydration with formic acid as a catalyst.
The processes are outlined in the diagram below:
The table below explains the function of the different processes involved in the production of furfural from PPF.
| Process | Materials | Function |
| Mechanical Comminution | - | To break down PPF into smaller pieces to increase the surface area to volume ratio |
| Ionic liquid Pre-treatment | 1-butyl-3-methylimidazolium chloride ([BMIM]CL) | To remove lignin from hemicellulose to allow for easier hydrolysis. |
| Acid Hydrolysis | Formic Acid | To break xylan polypeptide chains into xylose monomers |
| Dehydration | Formic Acid | To convert xylose monomers into furfural |
The process of acid hydrolysis and dehydration is shown in the reaction pathway below:
Chemical Intermediate
Furfural is an important renewable chemical feedstock solvent for the production of Furfuryl alcohol, Furoic acid, Furfurylamine, methylfuran, Furan and tetrahydrofurfuryl alcohol.Furoic acid, Furfurylamine, Methylfuran, Furan and tetrahydrofurfuryl alcohol. These chemicals have a wide variety of uses in the chemical industry for example flavour agents and resin.
Furfural is used as a solvent for refining of lubricating oils, butadiene, rosin, and other chemicals; solvent for nitrocellulose, cellulose acetate, shoe dyes. It is also used as a solvent in petrochemical refining to extract dienes from other hydrocarbons. Furfural acts a reactive and is a good wetting solvent for the manufacture of abrasive wheels, brake linings and refractory products for the steel industry.
Other applications
Furfural with platinum catalyst, is used as an excellent catalyst for decarbonylation reactions.It is utilized in the production of high quality motor oil with improved  temperature-viscosity properties.Furfural is also used in the separation of aromatic, polar components, and mercaptans, that are removed from petroleum, by means of furfural extraction. It is used as a decolorizing agent to refine crude wood rosin.Furfural is used in the purification of an anthracene for the preparation of vat dyes, lacquers and rubbers.
temperature-viscosity properties.Furfural is also used in the separation of aromatic, polar components, and mercaptans, that are removed from petroleum, by means of furfural extraction. It is used as a decolorizing agent to refine crude wood rosin.Furfural is used in the purification of an anthracene for the preparation of vat dyes, lacquers and rubbers.
| PRODUCT IDENTIFICATION | ||
| CAS NO. | : | 98-01-1 |
| EINECS NO. | : | 202-627-7 |
| FORMULA | : | C4H3OCHO |
| MOL WT. | : | 96.09 |
| H.S. CODE | : | 2932.12 |
| Volaties Maximum as per IS 101-1964 | : | 1% |
| SYNONYMS | : | 2-Formylfuran; 2-Furanaldehyde; 2-Furancarbonal; Cyclic aldehyde; 2-Furaldehyde; 2-Furaldehído; 2-Furaldéhyde; alpha-Furole; Artificial ant oil; Fural; |
| PHYSICAL AND CHEMICAL PROPERTIES | ||
| PHYSICAL STATE | : | Clear to light brown liquid |
| ODOUR | : | odourless |
| ODOUR | : | odourless |
| MELTING POINT | : | -36 ~ -39 °C |
| BOILING POINT | : | 162 – 167 °C |
| VAPOUR PRESSURE | : | 37 – 39 mm at 100 °F |
| DENSITY | : | 11.60 – 11.72 lbs/gal |
| SPECIFIC GRAVITY | : | 1.16 |
| SOLUBILITY IN WATER | : | 8 (g/100g) |
| VAPOR DENSITY | : | 3.3 |
| DEXTROSE EQUIVALENT | : | 40 – 45 % |
| DEXTROSE EQUIVALENT | : | 40 – 45 % |
| DRY SOLIDS | : | 86 % |
| MOISTURE CONTENT | : | 13 – 15 % by weight |
| MINIMUM TOTAL SOLIDS | : | 85 % |
| ACIDITY | : | 0.6 ml max |
| MAXIMUM SULPHATED ASH | : | 0.3 % |
| pH | : | 4.8 – 5.2 |
| MAXIMUM SULPHUR DIOXIDE | : | 400 |
| MAXIMUM ARSENIC PPM | : | 1.00 |
| MAXIMUM COPPER PPM | : | 5.00 |
| MAXIMUM LEAD PPM | : | 2.00 |
| STABILITY | : | Stable under ordinary conditions. Light , air sensitive. |
| SALES SPECIFICATION | ||
| APPEARANCE | : | clear to pale yellow liquid |
| PURITY | : | 98.5% min |
| ACIDITY | : | 20 max (mEq/l) |
| MOISTURE | : | 0.2% max |
| REFRACTIVE INDEX | : | 1.525 – 1.527 |
| FINAL BOILING POINT | : | 170 °C |
 TDS Specification TDS Specification |
 PDF MSDS PDF MSDS |
|---|